ATP molecule in an enzyme's active site, illustration
Bildnummer 12539265
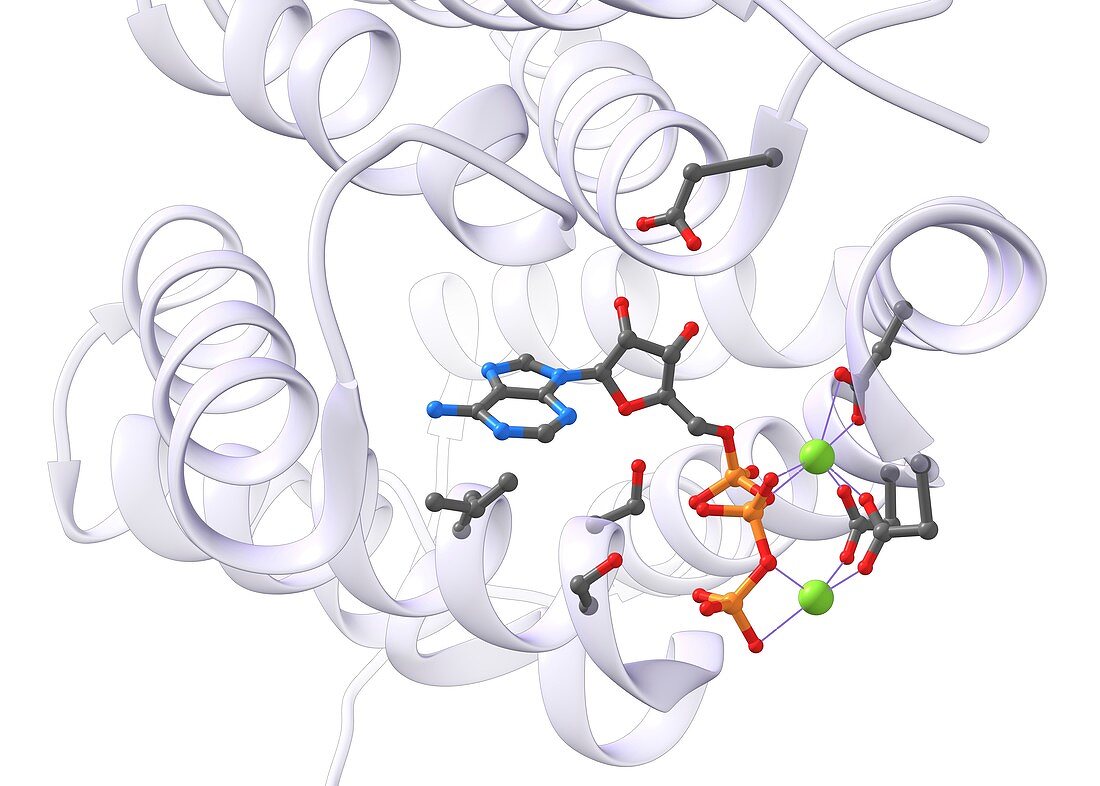
| Illustration of a model of ATP (adenosine triphosphate) in its bonding site in one of the kinase enzymes of glycolysis. The magnesium ions (Mg(2+), green) are held in place by electrostatic interactions (purple) between the triphosphate oxygen atoms and the carboxyl groups of several aspartic acid residues. Hydrogen bonds to the sugar and the phosphate group are also important, as is the dispersion interaction between the adenine electron cloud and an isoleucine side-chain. The Mg(2+) cations stabilize the cumulative negative 4 charge of the ATP triphosphate group. They also activate ATP as a phosphorylating agent, stabilizing the negative charge makes ATP a better substrate for nucleophilic attack, and it makes ADP a better leaving group. | |
| Lizenzart: | Lizenzpflichtig |
| Credit: | Science Photo Library / GREG WILLIAMS |
| Bildgröße: | 6995 px × 4996 px |
| Modell-Rechte: | nicht erforderlich |
| Eigentums-Rechte: | nicht erforderlich |
| Restrictions: | - |
Preise für dieses Bild ab 15 €
Universitäten & Organisationen
(Informationsmaterial Digital, Informationsmaterial Print, Lehrmaterial Digital etc.)
ab 15 €
Redaktionell
(Bücher, Bücher: Sach- und Fachliteratur, Digitale Medien (redaktionell) etc.)
ab 30 €
Werbung
(Anzeigen, Aussenwerbung, Digitale Medien, Fernsehwerbung, Karten, Werbemittel, Zeitschriften etc.)
ab 55 €
Handelsprodukte
(bedruckte Textilie, Kalender, Postkarte, Grußkarte, Verpackung etc.)
ab 75 €
Pauschalpreise
Rechtepakete für die unbeschränkte Bildnutzung in Print oder Online
ab 495 €
